ISSN: 2206-7418
Nanotheranostics 2017; 1(3):261-271. doi:10.7150/ntno.18964 This issue Cite
Research Paper
The biocompatibility studies of polymer dots on pregnant mice and fetuses
1. Key laboratory of Optoelectronic Devices and System of The Ministry of Education/Guangdong Province, College of Optoelectronic Engineering, Shenzhen University, Shenzhen Guangdong Province 518060, China;
2. Key laboratory of Biomedical Engineering of Shenzhen, College of medicine, Shenzhen University, Shenzhen, Guangdong 518060, China;
3. Shenzhen Key Laboratory of Marine Bioresource and Eco-environmental Science, Shenzhen Key Laboratory of Microbia Genetic Engineering, College of Life Sciences and Oceanography, Shenzhen University, Shenzhen 518060, China;
4. Department of Biomedical Engineering, Southern University of Science and Technology, Shenzhen, Guangdong 518055, China;
5. State key laboratory on integrated Optoelectronics, College of Electronic Science and Engineering, Jilin University Changchun, Jinlin 130012, China;
6. School of Electrical and Electronic Engineering, Nanyang Technological University, Singapore 639798, Singapore.
Abstract
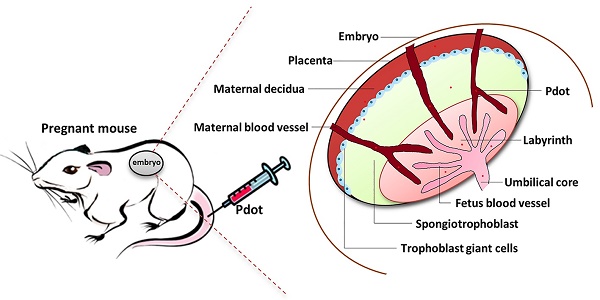
Semiconducting polymer dots (Pdots) are small nanoparticles consisting primarily of fluorescent pi-conjugated polymers which show superior optical properties for biological imaging and biosensors. It is necessary to explore systematically the toxicity of Pdots on animals before extensive biomedical applications. The reproductive system is very sensitive to the external invasion and essential for species reproduction as well. In this work, we used the pregnant mice to investigate the reproductive toxicity of Pdots. The changes in body weight of each maternal mouse were recorded every two days. The main organs were collected and analyzed as soon as all the pregnant mice were sacrificed on the 15th embryonic day. Distributions of Pdots on maternal major organs and tissues were examined in frozen tissue sections. Hematoxylin and eosin (H&E) staining was performed to investigate the histopathological changes of maternal organs. The blood chemistry test was applied to study the effects of Pdots on organ functions. Female hormones were evaluated by immunoassays. The amniotic fluid was inspected for assessing their penetration ability of Pdots. Levels of placenta growth related factors were detected by RT-PCR to evaluate the function of placenta. These results showed that Pdots were mainly accumulated in liver and spleen, and no apparent impact was observed on maternal body weight and organs coefficients. Histopathological images also showed normal tissue morphology compared with the untreated group. The female hormones levels did not show significant difference among the three groups as well. Trace amount of Pdots could get into the amniotic fluid but did not change the placental functions and the early development of fetus. Our results demonstrated that Pdots have excellent biocompatibility and no reproductive toxicity under the dosages used in this work, which means that Pdots have great potential in preclinical applications in the future.
Keywords: nanoparticles, polymer dot, reproductive toxicity.

 Global reach, higher impact
Global reach, higher impact