ISSN: 2206-7418Nanotheranostics
Nanotheranostics 2017; 1(1):1-22. doi:10.7150/ntno.17109 This issue Cite
Review
Remotely Triggered Nano-Theranostics For Cancer Applications
University of Massachusetts Lowell, Department of Chemical Engineering, Francis College of Engineering, 1 University Ave, Lowell, MA 01854, USA.
Abstract
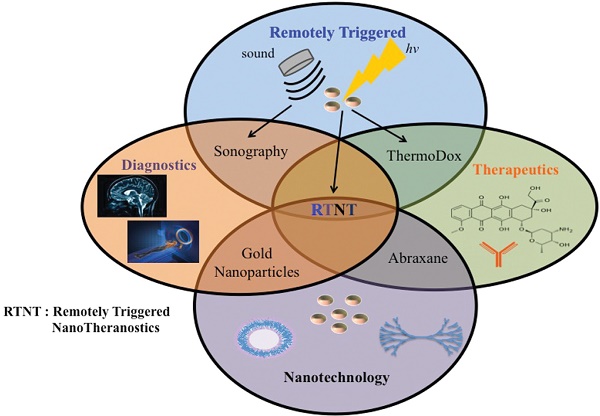
Nanotechnology has enabled the development of smart theranostic platforms that can concurrently diagnose disease, start primary treatment, monitor response, and, if required, initiate secondary treatments. Recent in vivo experiments demonstrate the promise of using theranostics in the clinic. In this paper, we review the use of remotely triggered theranostic nanoparticles for cancer applications, focusing heavily on advances in the past five years. Remote triggering mechanisms covered include photodynamic, photothermal, phototriggered chemotherapeutic release, ultrasound, electro-thermal, magneto-thermal, X-ray, and radiofrequency therapies. Each section includes a brief overview of the triggering mechanism and summarizes the variety of nanoparticles employed in each method. Emphasis in each category is placed on nano-theranostics with in vivo success. Some of the nanotheranostic platforms highlighted include photoactivatable multi-inhibitor nanoliposomes, plasmonic nanobubbles, reduced graphene oxide-iron oxide nanoparticles, photoswitching nanoparticles, multispectral optoacoustic tomography using indocyanine green, low temperature sensitive liposomes, and receptor-targeted iron oxide nanoparticles loaded with gemcitabine. The studies reviewed here provide strong evidence that the field of nanotheranostics is rapidly evolving. Such nanoplatforms may soon enable unique advances in the clinical management of cancer. However, reproducibility in the synthesis procedures of such “smart” platforms that lend themselves to easy scale-up in their manufacturing, as well as the development of new and improved models of cancer that are more predictive of human responses, need to happen soon for this field to make a rapid clinical impact.
Keywords: nanomedicine, oncology, externally-triggered, image guided, photoactivated.

 Global reach, higher impact
Global reach, higher impact