ISSN: 2206-7418Nanotheranostics
Nanotheranostics 2020; 4(4):233-241. doi:10.7150/ntno.46928 This issue Cite
Research Paper
Electrostatic reaction for the detection of circulating tumor cells as a potential diagnostic biomarker for metastasis in solid tumor
1. Institue of Reproductive Health, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, Hubei, China.
2. Department of Urology, the First Affiliated Hospital of Xiamen University, Xiamen 361003, Fujian, China.
3. Clinical Trial Management Platform, Jinhua Municipal Central Hospital, Jinhua 321000, Zhejiang, China.
4. Department of Clinical Medicine, Fujian Medical University, Fuzhou 350005, Fujian Province, China.
Abstract
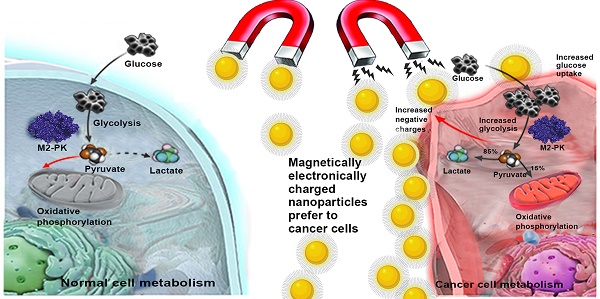
The detection of circulating tumor cells (CTCs) from blood samples is important to predict metastatic spread of cancer cells. However, effective quantification and identification of CTCs in solid tumors remain a challenge. Aerobic glycolysis is a hallmark of cancer cells, which makes cancer cells have more negative membrane potentials than that of normal cells. Herein, we reported a CTC isolation method with 80.7% capture efficiency based on electrostatic reaction, which was accomplished within 30 min in mimic clinical samples. Following in vitro verification using Lewis lung carcinoma (LLC1) (EpCAM-positive) and B16F10 (EpCAM-negative) melanoma cells, syngeneic tumor models were used to evaluate specificity and sensitivity of the surface charged nanoparticles. After subcutaneous implantation, blood was drawn from mice every three days, and CTCs were successfully detected in all implanted subjects. From 100 µl blood samples, the minimum amount found in blood was 9-34 CTCs on 3 day and the maximum was 94-107 CTCs on 15 day. Besides, the isolated CTCs cells remained viable and verified by re-implantation. This study confirms that our multifunctional nanoparticles are highly efficient in detecting CTCs in tumor metastasis and has huge potential in translational medicine.
Keywords: Circulating tumor cells (CTCs), Syngeneic tumor models, Nanoparticles, Bioelectricity
